How do the organs of our body develop and how are their physiological functions controlled? Are energy homeostasis, innate immunity and ageing linked to each other? Do fasting or overeating affect our immune system? What role does the gut microbiome play? To address these and other questions, we utilize the genetic model systems fruit fly, mouse and zebrafish in combination with biochemistry and molecular cell biology.
The overall goal of our research is to achieve a greater understanding of the complex links between metabolism and disease. The focus of our work is on lipid metabolism and its essential roles in energy homeostasis. We want to elucidate on a molecular basis how changes of energy homeostasis affect development and transition to pathological phenotypes.
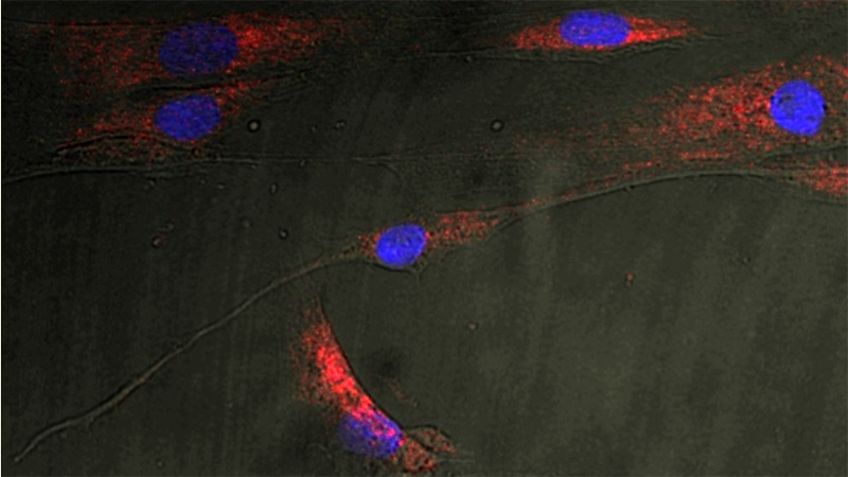
To address our questions we use genetic model systems fruit fly, mouse and zebrafish (e.g. genomic engineering and the latest technologies of genome editing) and in vitro techniques such as siRNA-mediated “knock-down” in combination with molecular biology, protein- and lipid-biochemistry, cell biology, as well as microscopical techniques.
Areas of research
- Regulators and regulation of (sphingo) lipid metabolism and body fat homeostasis with focus on ceramide synthases (CerS), highly conserved key enzymes in the lipid metabolism of eukaryotes
- Study of peroxisome and lysosome biogenesis
Ceramide Synthases (CerSs)
Studies on enzymes in the ceramide-synthesizing pathway indicate that the pathway serves as an important sensor of the lipid status. We identified the Drosophila CerS enzyme Schlank as modulator of transcriptional response. CerS Schlank fulfills a double role as enzyme and nuclear, transcriptional regulator sensing lipid levels and transducing the information to the level of gene expression. The role as transcriptional regulator is mediated via the homeodomain within the CerS enzyme.
For an efficient analysis of the role of CerS as transcriptional regulator and as enzyme we established a “genomic engineering” system. This system allows genetic manipulation of the Schlank gene into almost any desired mutant allele. We have combined these alleles with flippase (Flp) recognition target (FRT) sites to render it excisable by Flp-activity enabling us to perform a great variety of genetic manipulations including tissue-specific allele switching and the generation of genetic mosaics in post-mitotic or polyploid cells at any time and in a tissue-specific manner.
Equipped with these unique tools we analyse the functions of CerS Schlank, an enzyme doing double duty as transcriptional regulator, in development and pathogenesis and in the background of disease models.
Since the functions of CerS are conserved, we transfer the knowledge derived from our Drosophila model (e.g. knowledge on critical motifs for transcriptional regulation or enzymatic activity) into the mammalian mouse model.