Forschung
New tool compounds to study metabolic flexibility & plasticity in cancer organoids
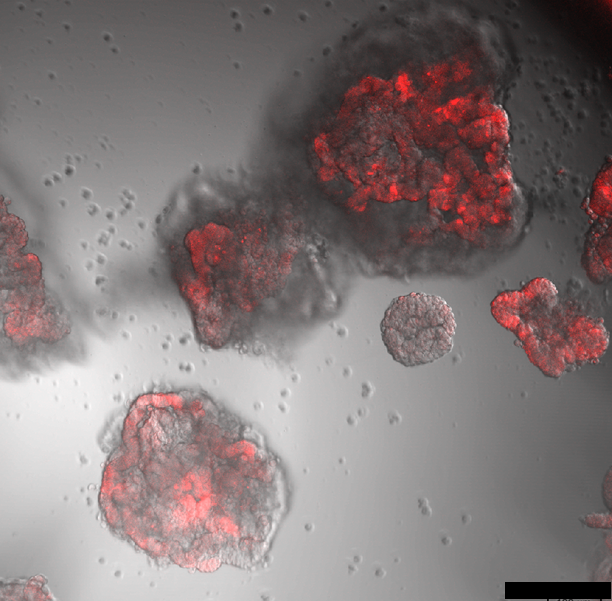
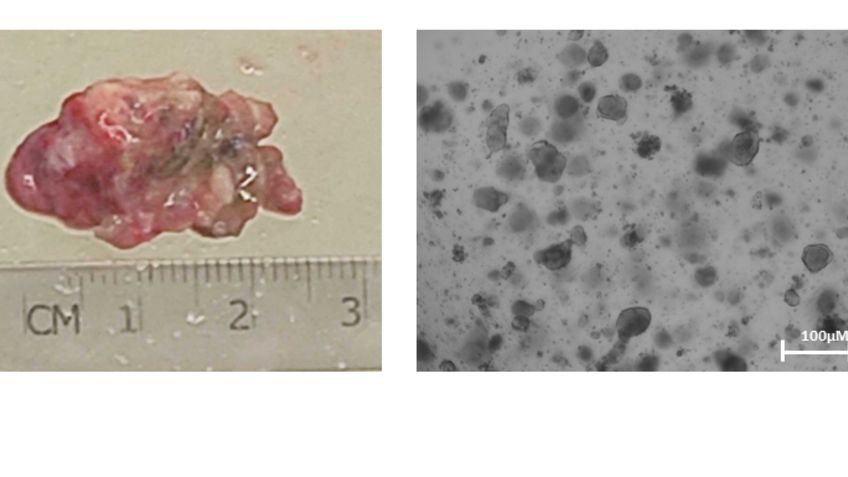
Deregulation of metabolism is a recognized hallmark of cancer. The flexibility and plasticity of cancer to adapt their metabolism to environmental changes are known contributors to chemoresistance. However, we do not have a full picture of the intrinsic and extrinsic factors that determine the metabolic adaptability of a cancer cell. Understanding the metabolic regulation of cancer might enable the development of novel treatment strategies to combat chemoresistance development. The Reckzeh lab uses patient-derived colorectal cancer organoids with an extremely adaptable metabolism to screen for small molecules that target key metabolic regulators which directly interfere with the metabolic adaptation potential. We use chemical structure optimization to derive tool compounds that we use to study the metabolic flexibility in cancer, chemoresistance development and ultimately provide potential treatment opportunities for patients.

Intestinal bioavailability of nutrients
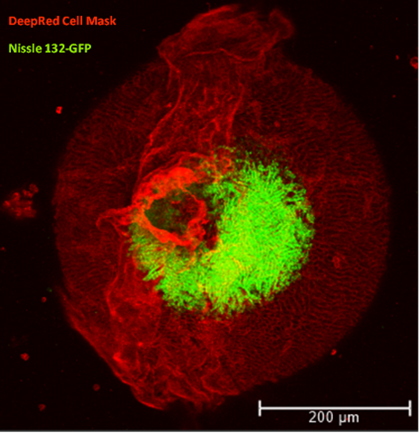
The majority of nutrients from consumed food are absorbed in the small intestine. Bioavailability of nutrients is influenced by neuronal and hormonal stimuli as well as the microbiome. The Reckzeh lab develops complex intestinal organoid models to study extrinsic and intrinsic stimuli that interfere with or induce the absorption of specific nutrients within the small intestine using co-cultures and chemical screens. The developed tool compounds will enhance our understanding of nutrient bioavailability and explore alternative strategies to prevent or treat malabsorption-associated and metabolic diseases, such as diabetes.